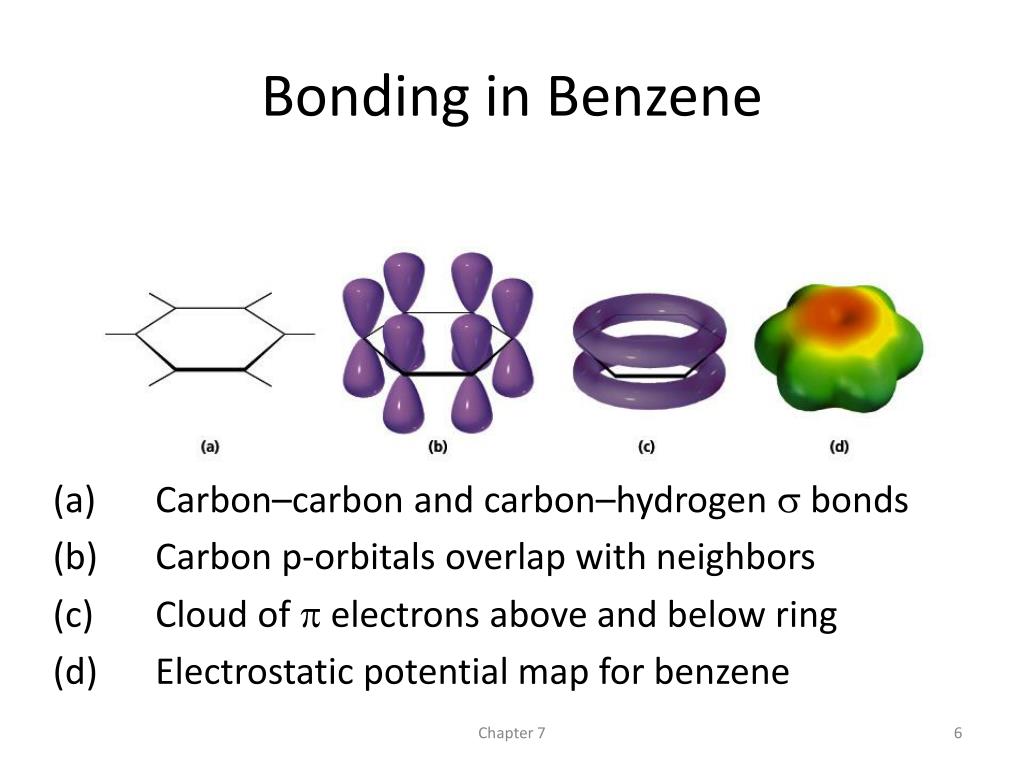
This involves 24 electrons in bonds formed by overlap of sp 2 hybrid orbitals on the carbon atoms with 1s orbitals on each hydrogen or with other sp 2 hybrids on other carbons. As in the case of ozone, we can treat the sigma bonds of benzene in valence-bond terms, dealing only with pi bonding by the molecular-orbital, method. The charge density of aromatic compounds is diffuse and delocalized such that. Notice, for example, the difference in acidity between phenol and cyclohexanol. The ChemDoodle Web Components library is a pure JavaScript chemical graphics and cheminformatics library derived from the ChemDoodle application and produced by iChemLabs. benzene (6 site), benzene (9 site), nitrobenzene, toluene (10 site). If you want to pause the game, just click the canvas again. The right paddle is controled by the computer. Start the game by clicking on the canvas. Resonance effects involving aromatic structures can have a dramatic influence on acidity and basicity. This is an example of a plugin written for the ChemDoodle Web Components library. A new advanced CIP descriptor engine is implemented for the most accurate and consistent CIP assignments and the MDL/BIOVIA stereochemical drawing interpretation algorithm is now implemented along side the IUPAC specification. 7.10: Delocalized Electrons Can Affect the Product of a Reaction. 11.13.0 JanuExecutive Summary ChemDoodle 2D v11.13.0 is a feature update wrapping up our latest massive work on stereochemistry. 7.8: A Molecular Orbital Description of Stability. This indicates that all C-C bonds are equivalent and intermediate between a single and a double bond. 7.9: How Delocalized Electrons Affect pKa Values. Which of the following has delocalized electrons chemdoodle delocalized benzene. As described in "Resonance," benzene can be represented by the resonance hybrid The presence of delocalised electrons is a hallmark of aromatic compounds. \) c.Īnother important molecule to which MO theory can be applied usefully is benzene.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
